  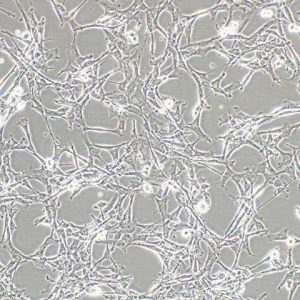
UCB samples were diluted in a proportion of 1:1 in a phosphate-buffered saline solution (PBS) and submitted to two different mesenchymal culture cell procedures: a) Method without Ficoll-Paque density gradient (d=1.077 g/mL) (Amershan Pharmacia) b) Method with Ficoll-Paque density gradient. Blood was collected in sterile syringes containing heparin sodium (1000 IU). Free written informed consent was obtained from the mothers or the next of kin following the guidelines of the local Institutional Review Board and Ethics Committee, according to certificate Nº 168/2005. Ten samples of UCB were collected from full-term placenta of healthy women, nonsmokers, nondrinkers, age ranging from 20 to 40 years, regardless ethnic group at the Obstetric Service of Hospital de Base Regional Medical School Foundation (FUNFARME) from the Medical School of São José do Rio Preto. 
This protocol was compared to the conventional procedure using Ficoll-Paque density gradient centrifugation in order to isolate mononuclear cells. These cells correspond to all nucleated cells present on UCB and not only to the low-density mononuclear cells isolated by the Ficoll-Paque density gradient. In face of these difficulties, our group has developed a mesenchymal stem cell culture of UCB from the isolation of nucleated cells present in the buffy coat collected through centrifugation without Ficoll-Paque density gradient. This system has proven to be more efficient and faster in separating mononuclear cells when in comparison to the conventional technique which uses Ficoll-Paque density gradient however, the release of this system for clinical practice yet requires further studies. Also, easy to handle and closely sealed filtration systems to concentrate mononuclear cells, the so-called SCF SYSTEM (the stem cell collection filter system), have already been tested. Moreover, these procedures also involve several phases of the manipulation process that increase the risk of contamination. Despite the efficacy of these procedures, the release of these products for clinical practice depends on the approval of the centers for disease control in several countries. In attempting to speed up the process making the mononuclear cell isolation processes faster, several methods have already been tested such as the use of poligeline, hydroxyethyl starch gel (HES) and gelatin to deplete red blood cells (RBC). This procedure increases the risk of contamination of UCB. Īll mesenchymal stem cell protocols of umbilical cord blood start with an isolation phase of mononuclear cells with Ficoll-Paque density gradient, which requires several manipulation and centrifugation phases of umbilical cord blood. Despite the positive outcomes observed in these groups, many studies have reported great difficulty to culture and maintain these cells in vitro, and others report a total failure in isolating and culture of these same cells. 
Mesenchymal stem cells different isolation and culture protocols of umbilical cord blood have already been employed successfully, all involving one early stage of mononuclear cell separation using Ficoll-Paque density gradient. Nevertheless, as these cells correspond to only a small portion of mononuclear cells present in each sample, it is necessary to isolate and multiply them in vitro. These characteristics create expectations of using cellular therapies to regenerate tissues and organs affected by the so called incurable diseases, such as neurologic, cardiac, and kidney diseases, etc. MSCs are capable of multilineage proliferation and differentiation, similarly to those observed in bone marrow cells. In the last decade, a considerable number of studies have proven that human umbilical cord blood (UCB) has hematopoietic stem cells (SCH) and a pool of mesenchymal stem cells (MSC). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
